Clinical studies are an essential step in the development of novel treatment methods for various diseases. They show researchers what works for the well-being of patients and what does not. In addition to safety and effectiveness they also determine whether the side effects of a new treatment are acceptable and what quality of life for patients is associated with a new therapy.
For the past several years, there has been increasing discussion of what patient centricity means in clinical trials. Premise is that better study designs, approaches, and services that are focused on the needs and preferences of patients can improve clinical research participation and, therefore, help advance the development of medicine and medical devices. Yet, despite the focus on this concept, the general consensus is that much work remains to realize its potential.
Discussion
Survey of over 1,600 patients with chronic illnesses was made. Patients showed high degree of willingness to take part in trials, but had low degree of trial experience. Main barrier to take part in trials appeared to be lack of awareness. As survey concluded, most patients (61%) werent being invited to take part by their physicians.
Providing prospective patients with the right information to make a decision starts with understanding what drives patients to participate in a trial.
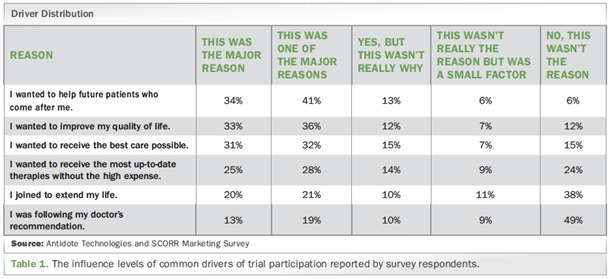
Based on the results of Antidote Technologies and SCORR Marketing survey project, respondents who had previously joined a clinical trial reported:
- For 75% major reason, or one of major reasons, was joining clinical trial to help future patients.
- 69% said they participated to improve their quality of life.
- 63% indicated they have had high motivation to participate in order to receive the best care possible.
The results of the aforementioned survey raise important points in regard to patient obstacles:
- Patients with life-threatening conditions are most concerned about gaining access to treatments.
- Individuals whose condition limits their mobility are concerned about logistical factors.
- People with asthma or allergies are concerned about the financial obstacles to participating in medical research. Those conditions tend to impact low-income people more.
No one-size-fits-all solution
More important than these findings is confirmation that there is not a one-size-fits-all solution for engaging individuals in medical research.
- It is important to consider the particular obstacles facing patients when they are making decisions about whether or not to partake.
- Patient-centric approaches to clinical trials may lead to effective recruitment and patient retention. Recruitment and retention of participants are key to the success of any clinical study. If either recruitment or retention falls short, a study may fail to achieve its objective. Researchers may be unable to answer the research question they posed and participants who were recruited to the study may have been placed at risk for no purpose.
- Thus, patient-centric approach may contibute to overcome obstacles in achieving clinical trial recruitment and retention goals. Antidote surveyed nearly 4,000 patients to find out what would make patients who take part in research feel like partners, not just participants. They asked the question directly: “What would make you feel like a partner in research?”. Across all conditions, talking with the doctors, clinical trial coordinators, and nurses involved in the trial was most important to patients.
- Researchers should put more effort upfront related to patients themselves. Focusing only on scientific endpoints and trial development can be a wasted effort if recruitment goals are not met. Also, if those enrolled don’t feel comfortable enough to stay in the trial.
- Patients favorable attitudes toward the value of research likely depend upon the presence of trusting relationships with their physicians and hospital.
- Taking the aforemention into consideration, patients can be partners in care, and hopefully someday, partners in finding the cure.
Conclusion
Patients are willing and able to take part in clinical trials for altruistic reasons, but are rarely given the opportunity to do so. Therefore, understanding and addressing patient needs and expectations are critical steps forward towards designing patient-centered trials rather than assuming that altruism alone will drive recruitment efforts. Although patient factors are only one aspect of conducting a successful trial there are ethical, scientific, and business reasons for taking a more patient-centric approach.
References:
1. Clinical trials from the patient perspective: survey in an online patient community, Pronabesh DasMahapatra, Priya Raja, Jeremy Gilbert & Paul Wicks, BMC Health Services Research volume 17, Article number: 166 (2017)
2. https://www.appliedclinicaltrialsonline.com (accessed April 15, 2021)
3. https://gcp.nidatraining.org/resources (accessed April 15, 2021)
4. https://www.antidote.me (accessed April 16, 2021)
5. Presenting the Importance of Patient Perspective in Clinical Trials, Eric T. Rosenthal, Oncology Times: May 25, 2005 – Volume 27 – Issue 10 – p 33-34
6. The Role of Patient Perspectives in Clinical Research Ethics and Policy: Response to Open Peer Commentaries on “Patient Perspectives on the Learning Health System”, Maureen Kelley, Cyan James, Stephanie Alessi Kraft, Diane Korngiebel, Isabelle Wijangco, Steven Joffe, Mildred K. Cho, Benjamin Wilfond, and Sandra Soo-Jin Lee, Am J Bioeth. 2016 Feb; 16(2): W7–W9




